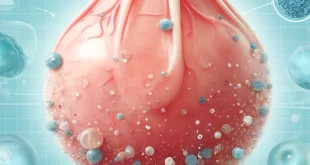
Introduction Scientists have discovered significant concentrations of microplastics in human testicular tissues, raising concerns about …
Read More »FDA Advancing Pathogen Reduction Technologies Fellowship
Three research opportunities are currently available in the Office of Blood Research and Review (OBRR) at the Center for Biologics Evaluation and Research (CBER), Food and Drug Administration (FDA) in Silver Spring, Maryland. Transfusion-transmitted infections (TTIs), while rare, remain a significant risk of blood transfusion. Although advances in donor screening and laboratory testing have decreased this risk, emerging …
Read More » medjouel.com Study Non Stop
medjouel.com Study Non Stop